The FDA has approved Eli Lilly’s Alzheimer’s drug donanemab, branded as Kisunla, for early symptomatic adults, providing a new treatment option for nearly 7 million Americans affected by the disease.
The US Food and Drug Administration (FDA) has announced the recall of 135 batches of potassium chloride extended-release capsules due to potential risks of cardiac arrest.
Prophet Premium Blends is recalling all Diamond Shruumz edible products nationwide due to toxic levels of muscimol, a chemical found in certain mushrooms, which have caused illness in 39 people across 20 states.
The US Food and Drug Administration (FDA) urged drugmakers and medical device manufacturers to improve racial and cultural diversity in clinical trials.
The Food and Drug Administration said that they have warned the Mexican-American bakery Bimbo Bakeries to stop using labels saying that their products may have potentially dangerous allergens when they contain none.
Coffee manufacturer Snapchill issued a countrywide product recall after federal regulators warned that its production process could produce a deadly toxin.
The FDA has approved the first menthol e-cigarettes for sale in the US, prompting criticism from parent groups who claim the agency has "failed American families."
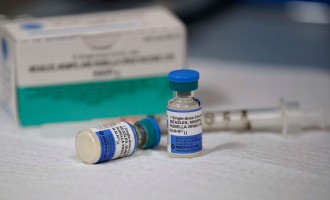
The Food and Drug Administration approved Merck’s new vaccine, Capvaxive, which was designed to protect adults from a bacteria known as pneumococcus that could cause serious illnesses like pneumonia, but is yet to face the CDC’s scrutiny.
The FDA has paused BioNTech's breast and lung cancer trial involving licensed treatment from Chinese biotech MediLink Therapeutics Ltd., citing safety concerns.
Neuroelectrics seeks FDA approval for its noninvasive head cap that uses mild electrical currents to treat brain diseases, potentially transforming care for epilepsy, depression, and Alzheimer’s.
An audit revealed that the FDA took over 15 months to act on a whistleblower complaint about conditions at an Abbott Nutrition factory.
The US FDA and DOJ have announced the formation of a task force to combat the illegal distribution and sale of electronic cigarettes, or e-cigarettes.
The Food and Drug Administration’s advisory committee endorsed Eli Lilly’s Alzheimer’s drug, donanemab, for approval to further slow the aggression of the specific type of dementia in patients despite criticisms regarding test patient deaths during clinical trials.
Abbott Laboratories announced that new FDA-cleared continuous glucose monitor sensors are available over-the-counter without a prescription.
The FDA is examining Diamond Shruumz-brand microdosing chocolate bars after eight individuals experienced symptoms including vomiting, seizures, and central nervous system depression, prompting concerns over the product's safety.
Subscribe to VCpost newsletter
Most Popular
- Nestle Cuts Sales Forecast as Shoppers Reject Price Hikes
- Social Security Payments Worth Over $4,800 To Go Out This Week; Here’s When You’ll Get Yours
- US Could See Another ‘Great Resignation’ as 3 in 10 Workers Plan To Quit in 2024: Survey
- Uber, Lyft Drivers Remain as Contractors After California Supreme Court Upheld Proposition 22
- Walmart Eyes $200 Million Investment in Autonomous Forklifts
- Maersk Agrees to Settlement with US Labor Department After Firing Whistleblower
- Delta’s CEO Flew to Paris for the Olympics While His Company Is Under Federal Investigation: Report
- Wiers Farm Recalls Produce Products Due To Potential Listerian Contamination